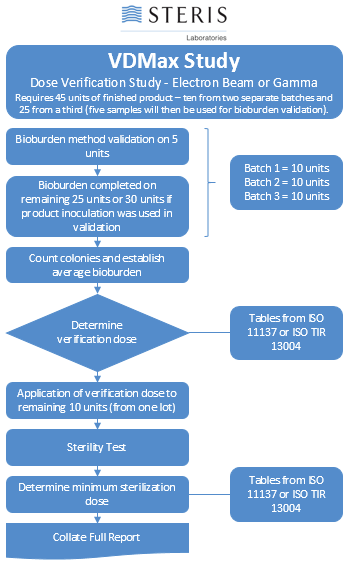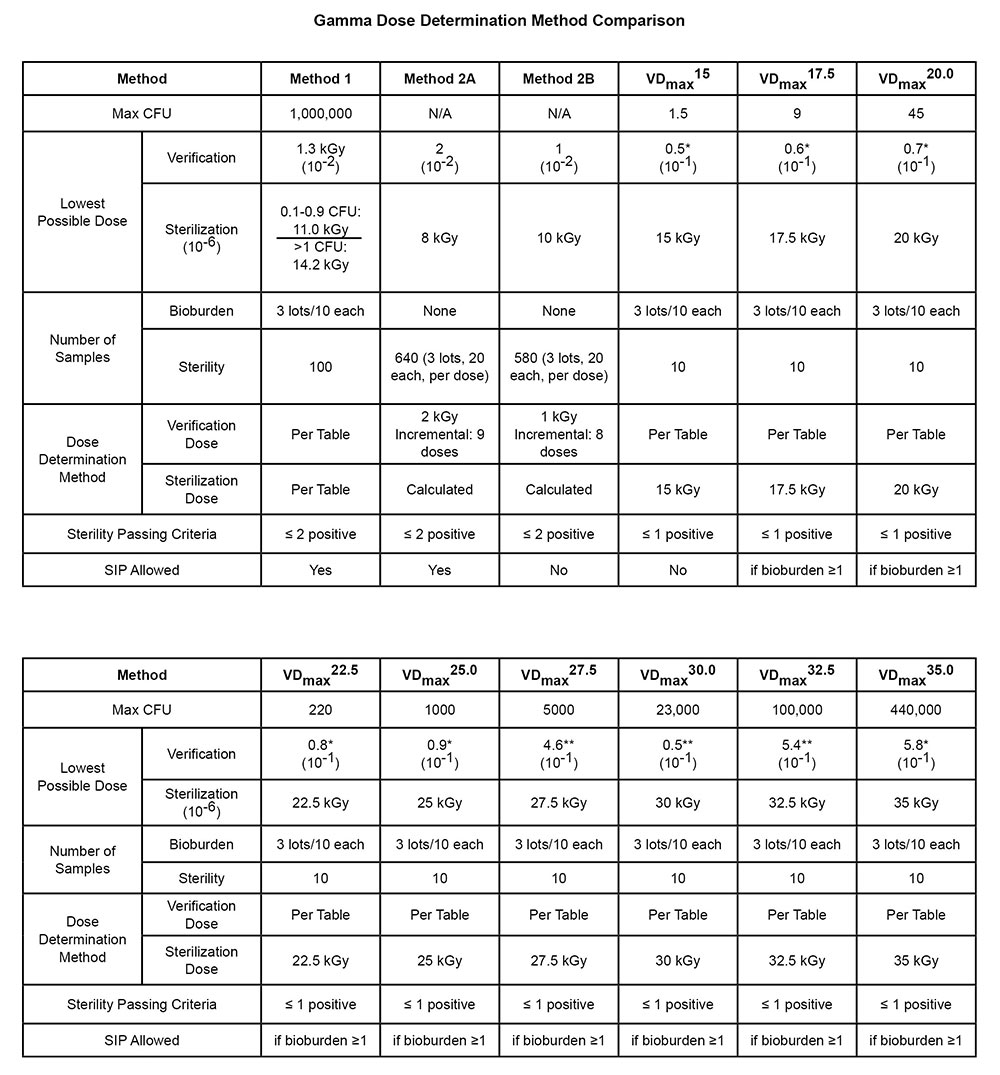
Comparison of AAMI Methods for Setting of Minimum Sterilization Dose with Irradiation | TechTip | STERIS AST

BS EN ISO 11137-2:2015+A1:2023 Sterilization of health care products. Radiation Establishing the sterilization dose

PDF) Validation of 15 kGy as a radiation sterilisation dose for bone allografts manufactured at the Queensland Bone Bank: Application of the VDmax 15 method
UNE EN ISO 11137-2:2015 Sterilization of health care products - Radiation - Part 2: Establishing the

ISO 11137-2:2006 - Sterilization of health care products - Radiation - Part 2: Establishing the sterilization dose

Guide to Irradiation and Sterilization Validation of Single-Use Bioprocess Systems - BioProcess InternationalBioProcess International
40[85:AOOTRR]2.0.CO;2/asset/eefab0e4-c696-4c3f-b163-b6569c5dba92/assets/graphic/i0899-8205-40-1-85-t01.gif)
An Overview of the Revised Radiation Sterilization Standards | Biomedical Instrumentation & Technology

ANSI/AAMI/ISO 11137-2:2006 - Sterilization of health care products - Radiation - Part 2: Establishing the sterilization dose: Amazon.com: Books

ISO 11137-2:2013/Amd1:2022 - - Amendment 1: Sterilization of health care products - Radiation - Part 2: Establishing the sterilization dose - Amendment 1